VR training for pharma manufacturing — Schedule M, Annex 1, and beyond.
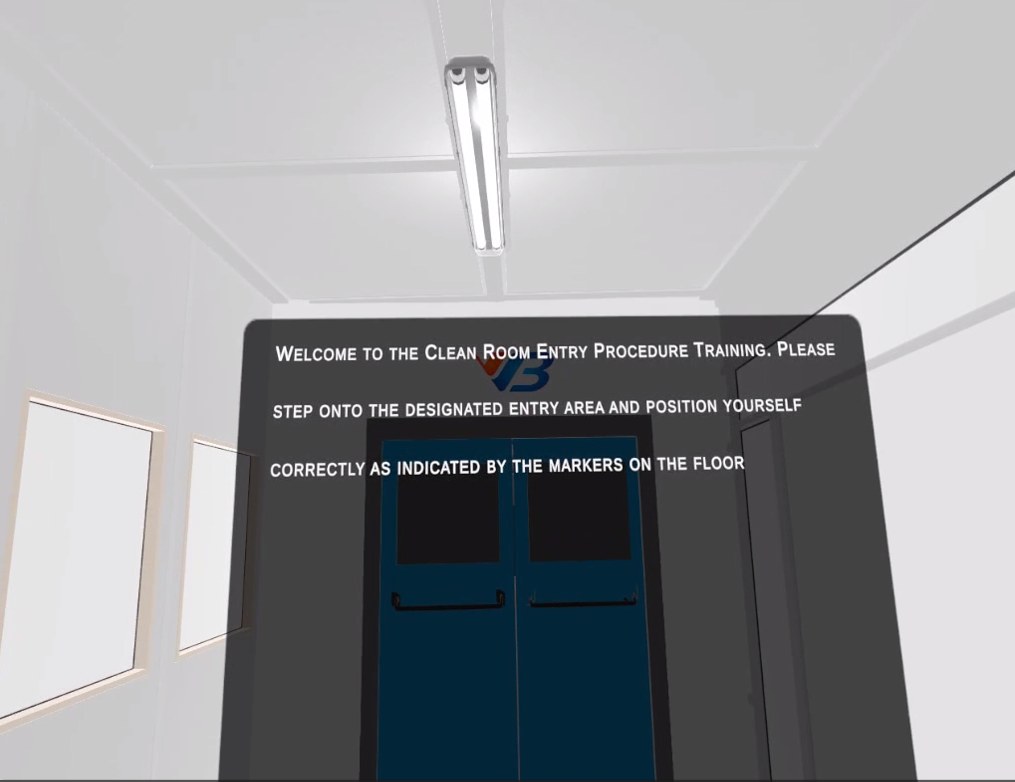
Drona VR for pharma manufacturing replicates your real SOPs step-for-step — cleanroom gowning, chemical handling, permit-to-work, electrical safety, LOTO. Built by VB Group EHS practitioners. Trusted at leading Indian pharma manufacturers.
Schedule M demands operator competency. Most pharma plants prove it on paper.
Revised Schedule M (effective 2024) requires demonstrable competency for every operator entering classified areas. Classroom training and SOP read-throughs no longer satisfy auditors. The shift is structural — but the training infrastructure inside most pharma plants has not caught up.
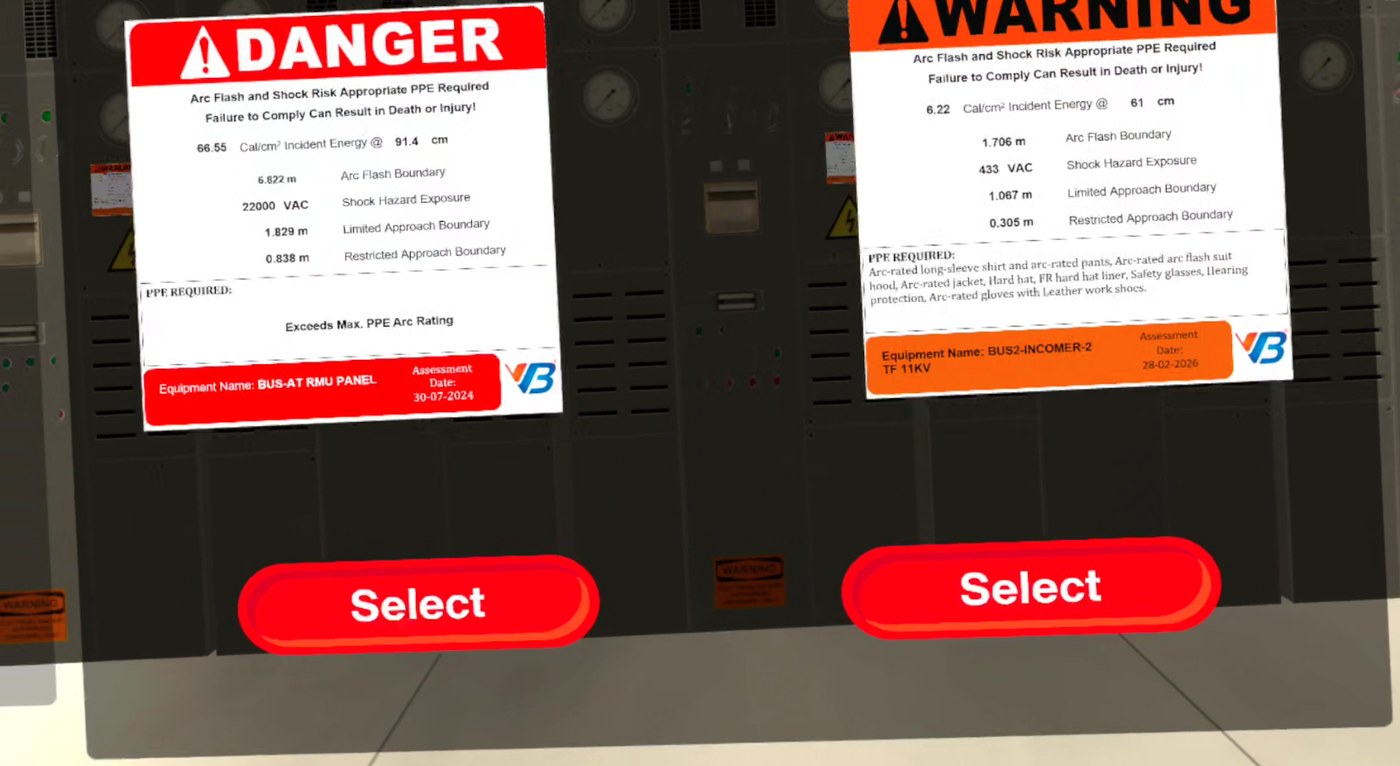
Cleanroom hygiene incidents are a Schedule M flag.
One contamination incident triggers an audit, a deviation report, and a CAPA. Operators trained on PowerPoint slides forget the gowning sequence in week three.
Onboarding takes lines offline.
New operators can only learn the SOP on the actual line. Production stops or new hires hover unproductively. Throughput suffers either way.
Audit documentation is a paper trail.
Trainers sign off attendance, not competency. Auditors increasingly demand evidence the operator can perform the SOP — not that they sat through training.
Deviation handling needs reflex, not recall.
When a cleanroom alarm triggers, the operator has seconds to follow the deviation procedure. Reflex is built through repetition — and you cannot repeat real cleanroom incidents.
Aligned with the standards your auditors check.
Every Drona VR pharma manufacturing module maps to the regulatory frameworks that govern your plant. Documentation is automatic, audit-ready, exportable.
Schedule M (Revised)
Indian pharma manufacturing licensing
EU GMP Annex 1
Sterile medicinal products
WHO GMP
Global pharma manufacturing
US FDA 21 CFR Part 211
Finished pharmaceuticals
ICH Q7
Active pharmaceutical ingredients
ISO 14644 / ISO 7-9
Cleanroom classifications
5 modules, engineered for pharma manufacturing plants.
Every module is ≤15 minutes, runs offline on the headset, and is engineered for eye health. Trainer cast view streams live to your EHS lead's screen.
Numbers from pharma deployments.
Representative outcomes from Drona VR deployments in pharma manufacturing. Specific numbers vary by plant and module.
The conversations we hear in every Pharma EHS office.
Our deviation log shows the same gowning errors quarter after quarter. Classroom training is not changing reflex.
The audit team is asking for evidence of competency, not attendance. We do not have a system that produces it.
I cannot take a line offline for two days every time we onboard new operators. We need a way to train them off-line, on real procedure.
We bought VR before. The content was generic. Our operators called it a game. We need training that respects the actual SOP.
Train pharma manufacturing for the work where mistakes are audit findings.
Book a 15-minute pharma demo. We will walk you through a relevant module live, with your SOP language.